
Pharmaceutical giants are pouring tens of millions of pounds into NHS services – including paying the salaries of medical staff and funding the redesign of patient treatment – as they seek to boost drug sales in the UK. Fortunately, this week, the Department of Health and Social Care (DHSC) has launched an open consultation on disclosing industry payments to the healthcare sector. We will now have an opportunity to have a say on whether manufacturers and commercial suppliers of medicines and devices should report details of the payments and other benefits they provide to healthcare professionals and organisations.

It would have been so much easier for us as patients if we had been able to trust that the medical profession was prescribing a drug because they believed it would be beneficial to us, and not because they were acting as big pharma salesmen.
Unfortunately, those within the medical profession do not wear their sponsors on their coats as racing drivers do, however, it has been common knowledge that big pharmaceutical companies have financial ties to them, but real transparency is lacking.
Drug firms are funding groups that lobby for greater investment in their disease areas, and in some cases are paying generous consultancy fees to influential healthcare professionals, including GPs who have worked as clinical leads for NHS England and have received as much as £480,000 each from industry since 2019. (source)
Drug companies are systematically funding grassroots patient groups that lobby the NHS medicines watchdog to approve the rollout of their drugs, says the Observer which carried out an investigation that found “of 173 drug appraisals conducted by the National Institute for Health and Care Excellence (Nice) since April 2021, 138 involved patient groups that had a financial link to the maker of the drug being assessed, or have since received funding.” (source)
Often, the financial interests were not clearly disclosed in Nice transparency documents, but the Observer did reveal that many of the groups that received the payments went on to make “impassioned pleas” to England’s medicines watchdog calling for treatments to be approved for diseases and illnesses including cancer, heart disease, migraine and diabetes. Others made submissions appealing Nice decisions when medicines were refused for being too expensive. (source)
Lack of Disclosure
One study published in the British Medical Journal (BMJ) in 2019 by Mandeville et al found that during NICE appraisal meetings, fewer than 25% of all relevant financial ties between patient organisations and pharmaceutical companies were disclosed. As discussed by Mandeville and colleagues, this lack of transparency increases the risk of conflicts of interest not being properly detected and managed.

Assessment at Englands National Institute for Health and Care Excellence Policy review
Undermining Credibility
Another study conducted Gentilini and Parvanova in 2020 assessed the relationship between UK-based patient organisation funding and companies’ commercial interests in rare and non-rare diseases. They found that companies predominantly funded patient organisations operating in therapeutic areas relevant to companies’ portfolios or drug development pipelines.

study of commercial determinants funding concentration and
disease prevalence
The researchers found that “the almost-perfect concordance between industry interests and patient organisation activities likely reflect the commercial attractiveness of conditions targeted by pharmaceutical companies, adding that “such close alignment between the interests of companies and patient organisations might undermine the credibility of patient organisations as perceived by the general public and might raise questions about patient organisations’ inputs in regulatory and health technology appraisals.
Important Future Research
The researchers made a very important point, saying that while their analysis “did not evaluate the effect of COVID-19 on the financial dynamics between pharmaceutical companies and patient organisations, we expect that the pandemic had
a substantial effect on the type, value, and distribution of payments. Future research should examine the impact of
COVID-19 on industry funding of patient organisations.” This is an area that certainly would be of interest to the general public.
The Public Has a Right to Know
Carl Heneghan and Tom Jefferson have reported that “The Independent Medicines and Medical Devices Safety Review, chaired by Julia Cumberlege, recommended that “the register of the General Medical Council (GMC) should be expanded to include a list of financial and non-pecuniary interests for all doctors. The public has a right to know.”
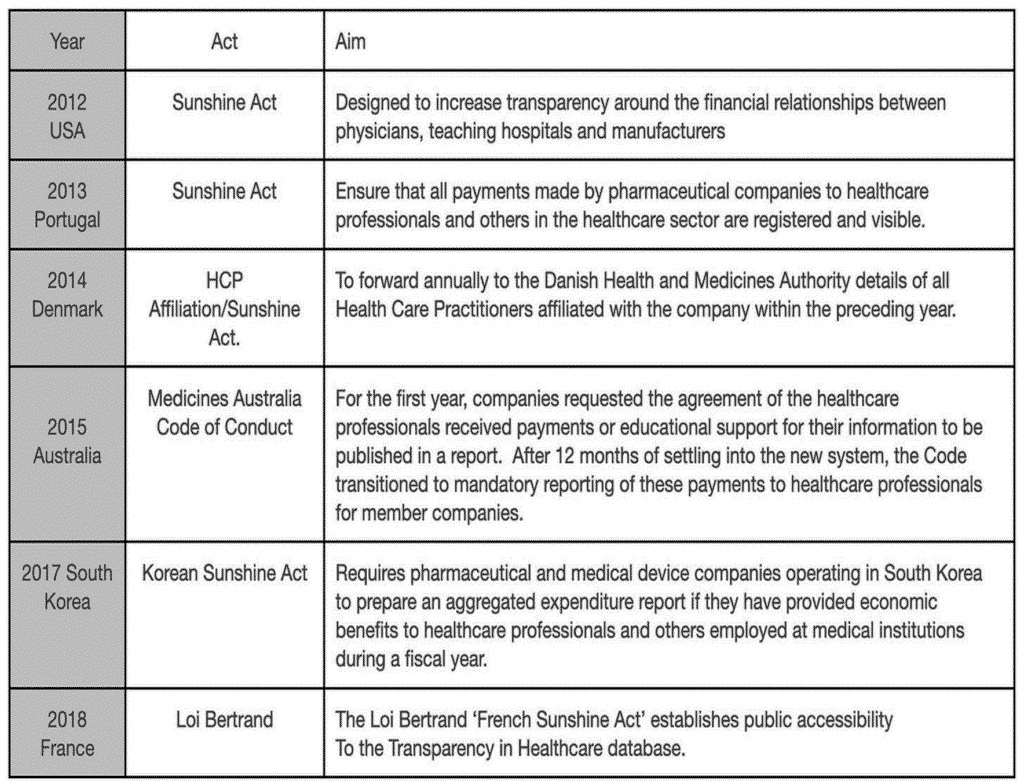
The House of Commons Health Select Committee recommended a GMC declaration register in 2005. Yet, unlike other countries that have thought this through and sorted the issue, the UK still lacks legislation. For example, The US Physician Payments Sunshine Act requires manufacturers to collect, track, and report all financial relationships with clinicians and teaching hospitals.
The Sunshine Act
Those with conflicts think the voluntary system works fine. However, the IMMDS review showed that the system is failing.
The Association of British Pharmaceutical Industries created a voluntary register, but because it was discretionary, vast numbers of payments to doctors were not disclosed.
NHS Trusts in England must also collate employees’ financial interests annually and publish them on their websites. In 2022, we asked how declarations of interest work – we found the system fails miserably.
In a random sample of NHS Trusts, three-quarters of registers did not routinely include all declaration of interest categories recommended by NHS England. In Scotland, only 14% of Boards published staff registers of interest. Organisations often had multiple registers lacking search facilities that were often uninterpretable.
In 2019, Carl told Sky News: “It’s really important that the public trust how we deliver medicines and no one really has an understanding of who is being paid what.
“There is evidence that conflict of interest distorts results and clinical practice because there is so much money in healthcare – there’s a real incentive for industry to get their product out there.”
One in Four Investigators Have Financial Ties
Financial and non-financial conflicts of interest are widespread among academic institutions and researchers. Industry sponsorship is often associated with restrictions on publication, lack of access to results and a shift in research emphasis. One in four investigators have industry ties, and roughly two-thirds of academic institutions hold equity in start-up companies that sponsor research at the same institutions.
If anything, the situation is getting worse.
The Royal Colleges have received more than £9m from drug and medical devices companies since 2015. Last month, the Observer reported a sharp rise in spending by drug firms in the UK health sector: “In 2022, the total value of such payments was more than £200m – up 26% compared with the previous year, and almost double the amount spent by industry in 2015.” From 2012 to 2016, the industry donated over £57m to UK patient organisations – the yearly sum more than doubled.
The new consultation highlights that the Health and Care Act 2022 allows the Secretary of State to impose a requirement for businesses to make public details of the payments to persons providing healthcare in the UK.
The consultation is open for six weeks and will close on 16 October 2023. The easiest way to respond is by completing the online survey. Views are being sought to define further who should be required to publish information – this is your opportunity to have a say. (source).
The Expose Urgently Needs Your Help…
Can you please help to keep the lights on with The Expose’s honest, reliable, powerful and truthful journalism?
Your Government & Big Tech organisations
try to silence & shut down The Expose.
So we need your help to ensure
we can continue to bring you the
facts the mainstream refuses to.
The government does not fund us
to publish lies and propaganda on their
behalf like the Mainstream Media.
Instead, we rely solely on your support. So
please support us in our efforts to bring
you honest, reliable, investigative journalism
today. It’s secure, quick and easy.
Please choose your preferred method below to show your support.
Categories: Breaking News

Yes down to the very last penny Nam & Shame
Yes, I agree Carol.
According to a friend of my younger brother, the $700 is nothing in comparison to the millions he has given to Ukraine. Why? We require it, she replied. Because vs02 we don’t know why, it must be it. We possess citizenship.
.
.
Click the link…………………………………………….https://m5.gs/SGRQYn
E glands a tiny place with a entire population less than most civilised countries major cities.
It irrelevant except for its covert global. Control.
Everyone knows they’re all corrupt. It’s not news
Not to you perhaps, but we have seen this last few years, not everyone knows they are corrupt. The news is we can now have a say at least.
Maybe tiny but probably the most expensive no matter what you want, a treatment or just a battery change in a watch. Just yesterday they offered 75£ for changing the battery in a Tag watch. The same cost me 12€ in Tenerife. For dental treatment or any type of screening we still go to Europe, our live saving drugs like antibiotics and painkillers just in case also comes from Europe.
Back to the topic, they are corrupt everywhere, that comes from the Rockafella system. Even in socialism and so called free health care you had to pay a fortune into a pocket to jump the list waiting for a heart surgery, if you couldn’t that was a death sentence. If you didn’t poisoned (vaccinated) your child by book the child was taken away. The whole system is corrupt, I don’t think we can change it unless we start with the root, but even then it takes a decade at least to have new docs with good knowledge.
I mean they must have known that this or that protocol (midazolam) was bad but they carried on killing for money. Who wants murderers and idiots (the vaxxed docs) for treatment?
Maybe tiny but probably the most expensive no matter what you want, a treatment or just a battery change in a watch.
Back to the topic, they are corrupt everywhere, that comes from the Rockafella system. Even in socialism and so called free health care you had to pay a fortune into a pocket to jump the list waiting for a heart surgery, if you couldn’t that was a death sentence. If you didn’t poisoned (vaccinated) your child by book the child was taken away. The whole system is corrupt, I don’t think we can change it unless we start with the root, but even then it takes a decade at least to have new docs with good knowledge.
I mean they must have known that this or that protocol (midazolam) was bad but they carried on killing for money. Who wants murderers and idiots (the vaxxed docs) for treatment?
“Three pharmaceutical firms ‘illegally boosted their profits’ with a scheme to hike the price of a UTI antibiotic prescribed millions of times by the NHS, market watchdog claims”
https://www.dailymail.co.uk/health/article-7062055/Four-pharma-companies-worked-inflate-drug-price-700-just-four-years.html
“In Greece, a life-transforming thyroid pill costs just £1 a month, but in Britain it’s £204 – so how can firms justify the drug daylight robbery?”
https://www.dailymail.co.uk/health/article-7076005/In-Greece-thyroid-pill-costs-1-month-firms-justify-drug-daylight-robbery.html
“Four pharma companies ‘worked together to fleece NHS of millions by inflating a drug price 700% in just four years”
https://www.dailymail.co.uk/health/article-7062055/Four-pharma-companies-worked-inflate-drug-price-700-just-four-years.html
The right question is that what the cause of the skyrocketing thyroid problems at the first place. Why drugging is needed.
Same with the rest.
They hide the most effective ‘drugs’, the antioxidants. They give bad advice on diet, they cause countless illnesses with ‘vaccinations’. They created the market for themselves.
Even if we can have a word, what’s the point? These docs were ‘raised’ on allopathic medicine are unable to cure a thing anyway.
Due to their extra high salaries and bribes (eg payment for given C19 vax) people who chose the profession chose it for money. Apart from surgeons and maybe 1-2 specialists they good for nothing and lazy to work anyway.
As a proud anti-vaxxer”, I know that every “vaccine” that has been put into people and animals, in all of history, is a toxic mix that only does harm to the victim.
There is not one single entity in “vaccines” that improves the health of the recipient: not even a B Vitamin or something that might do some good.
In fact the opposite of what the medical “profession” say is true, and these new mRNA Frankenshots have been proven beyond doubt to contain only substances and nanotech designed to maim, sterilize, kill or transhumanize the populations.
I am at the end of my tether with being lied to by MSM, and want Nuremberg 2.0, but I see nothing happening except bombardment by more wicked lies.
The vaccine-injured must come together to document and record everything being done to them: both individually and as a global group bring cases against the perpetrators.